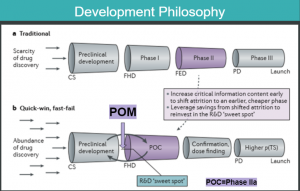
As part of our weekly business meetings, I present to the team information about some topic that the other intern and I have been assigned. One example of this was looking into the developmental model that EncepHeal Therapeutics partly mirrors. The Chorus Model, an internal branch of Eli Lilly, takes the “quick-win, fast-fail” approach to drug development, shown in Figure 1, vs. the traditional model. The purpose of presenting on this model was to share the aspects of Chorus that serve to expedite the pipeline of drug development, in addition to the ways EncepHeal Therapeutics can capitalize and expand on these strategies.
The Chorus model differs from the traditional model in that instead of obtaining their proof-of-concept (POC) at the end of phase II, which typically costs about $40 million, they get POC at an earlier point. They don’t invest in the later, more expensive and larger clinical trials until they have data providing some proof that their compound(s) are likely to be successful in a larger population of patients. This is called their “lean-2-proof-of-concept,” with the premise of getting as many early-stage compounds to POC as cost effectively as possible. They don’t invest in any manufacturing and commercialization of the product until they have a validated mechanism of action for their drug candidate. This higher probability of technical success saves them a lot of money if, let’s, say, that drug candidate would have otherwise failed at a later stage for a reason that wasn’t elucidated at an earlier point. EncepHeal Therapeutics is looking to follow this approach. They have several closely related compounds that are going through pre-clinical testing and will be refined into newer generation compounds if there isn’t sound proof of concept, such as a safety profile that will likely translate well into humans.
By presenting this model to the the CEO, CSO, and a couple of business and science advisors, EncepHeal members can identify areas where they would like to differ from the Chorus Model, to be even more efficient along their drug development pipeline. For example, one alternative strategy that was discussed was the possibility of testing the lead compound in a very small sample of non-human primates (NHP), shortly following rodent testing. For a drug to become FDA-approved, various toxicology and safety studies need to be conducted, using two different animals. NHPs are typically the higher-organism chosen, due to the high likelihood of results being translatable to humans. However, this testing is very costly. The idea of testing a promising compound in a smaller-than-normal sample would allow a quicker premature confirmation that the lead compound is likely to be successful at later stages.
The opportunity to research ways in which EncepHeal is similar and different than other companies gives me a keen perspective into what makes biotechnology companies succeed and fail. There are plenty of biotechnology companies that start out okay, but then fail and crumble. To ensure that EncepHeal is not only successful but also efficient in it’s drug development strategy, it’s crucial to understand how other similar companies are run. At our weekly meetings we can discuss how we are and should model some of these strengths. For example, the operational efficiency within the Chorus Model partly lies with them defining what their critical success factor (CSF) is from the start, so that if a compound falls short of certain thresholds, it’s discontinued.
Stay tuned to learn how EncepHeal Therapeutics is modelling various strengths characteristic of the Chorus Model, such as determining the minimum essential criteria needed for drug compounds to move forward.